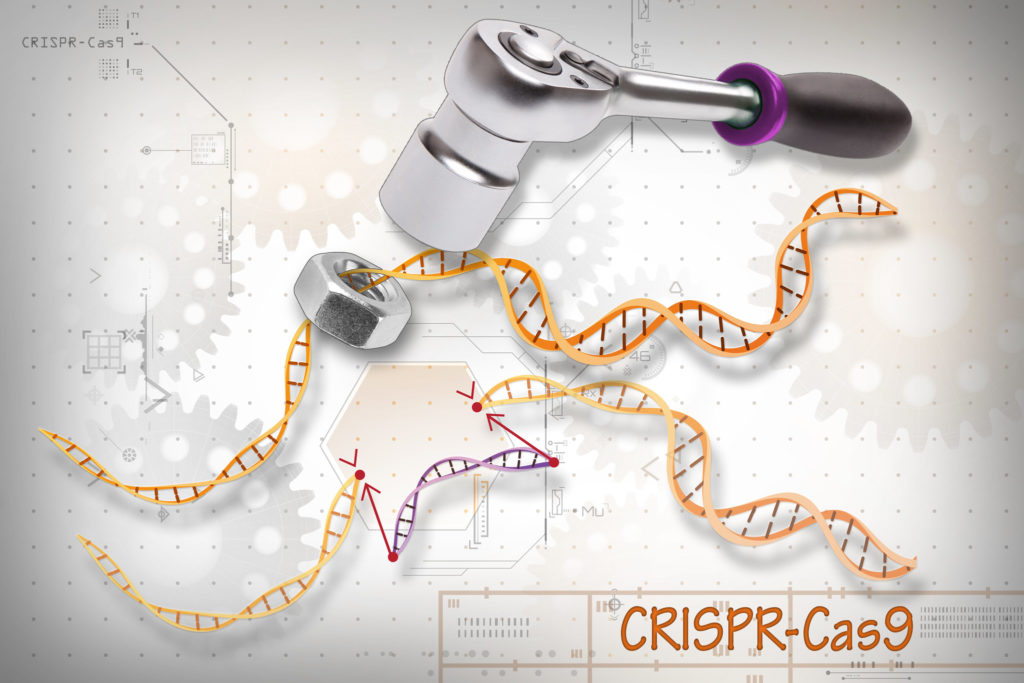
Stanford technology helps advance CRISPR-based cancer therapy
Since the discovery of powerful gene-editing tool CRISPR-Cas9, scientists and physicians have awaited the day it fulfilled its potential to improve human health. Now, researchers are one step closer to that goal.
Using CRISPR, which can add and remove bits of DNA in a cell, scientists at the University of Pennsylvania, Stanford and other institutions have reprogrammed immune cells to better target and fight cancer in three patients with advanced cancer. The therapy, which has been FDA approved, is one of the first in the United States to harness the power of gene editing to treat cancer, Stanford genome scientist Howard Chang, MD, PhD, told me.
The study, which appears in Science today, demonstrates that this type of CRISPR-based cancer therapy appears safe for patients so far and it could be an effective approach down the line.
Working with Carl June, MD, an immunologist at the University of Pennsylvania, Chang and his team supported the effort with technology built in his lab that sequences the RNA (or the in-use instructions for protein assembly) of single cells. The tool is based in something called “single-cell genomics,” which allows Chang and others to keep tabs on genomic changes in individual cells.
The trial’s approach takes a page from immunotherapy, in which a person’s own immune cells, specifically T cells, are removed, experimentally programmed to attack tumor cells, and then put back into the body.
Normally, these therapies rely on an external vehicle (like a neutralized virus) to deliver a special gene to the T cell. The difference here is that researchers are also editing the DNA in live cells using CRISPR to enhance their cancer-fighting abilities.
The T cells removed and edited using CRISPR are somatic cells, meaning they will not be passed onto future generations.
There are many different types of T cells, said Chang, a Howard Hughes Medical Institute investigator, and not all of them are genetically programmed to attack cancer cells. The goal is to use CRISPR to make more of the cancer-fighting variety, and then give them back to the patient.
Specifically, researchers target something on the cell known as a receptor, a protein that sits on the T cell’s surface and helps the T cell recognize other cells it’s meant to attack.
“Let’s say normally, there’s a T cell that’s involved in an allergic reaction to pollen,” said Chang. “We can use CRISPR to alter the cell so that it doesn’t react to pollen anymore, and instead, only fights cancer.”
This, in theory, is how it should work every time, but DNA editing is much messier in reality.
“It’s possible that you can make any combination of the changes you intend to make,” said Chang. In this case, the team wanted to make three edits, two to rid the cell of its natural receptors, and one to increase immune activity. But there’s only so much precision scientists can wield when editing biology on the micro-scale.
“You could end up with edits one and two, but not three. Or one and three, but not two, and so on,” said Chang. “It’s quite tricky to figure out which ones have stuck and which are missing.”
But in this process, knowing the success rate isn’t critical until later.
“There’s a time element here — we want to take the cells out of the patients, edit them, and put them back as soon as we can,” said Chang. For that reason, all of the edited cells, regardless of which changes stick, go back into the patient. T cells with faulty edits tend to die off, while those that successfully attack the cancer cells tend to flourish, Chang said.
After about three months, researchers take another sample from the patient, and that’s where Chang’s single-cell genomics tool comes into play.
“My lab has been interested in applying single-cell genomics directly to patient material and we’ve pioneered a series of technologies that not only does that, but does so in a way that’s more sensitive and on a large scale.”
In this study, Chang’s single-cell analysis tool allowed scientists to see exactly what changes CRISPR successfully made, how those changes altered the T cell molecularly and how those changes affected the cell’s function, providing key clues as to how the T cells are attacking the tumors.
“If you think of all of these edited T cells like they’re in a horse race, analyzing these cells is like being able to see which horse wins the race, but also that horse’s speed, gait, and all of the critical details that make that horse the best,” said Chang.
It’s still too early to name the therapy a success, but the preliminary results demonstrate that so far, CRISPR-based cancer therapy is safe and further investigation is warranted.
Image by Ernesto del Aguila III, National Human Genome Research Institute, National Institutes of Health
Popular posts
Category:

How viruses like the coronavirus can steal our sense of smell
Category:
What’s a virus, anyway? Part 1: The bare-bones basics