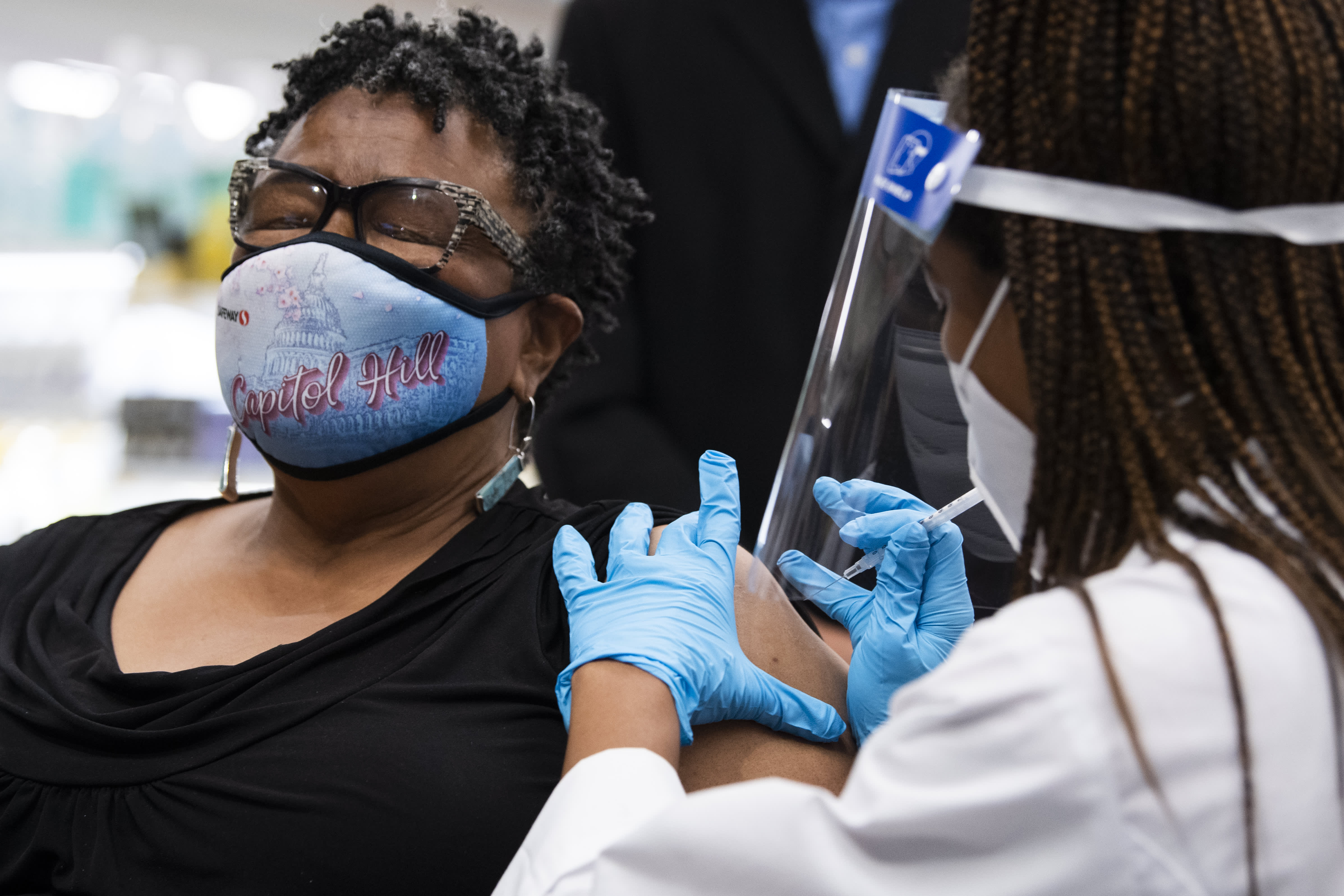
Germaine T. Leftwich, 67, receives a Pfizer covid-19 vaccine booster shot from Dr. Tiffany Taliaferro at the Safeway on Capitol Hill in Washington, D.C., on Monday, October 4, 2021.
Tom Williams | CQ-Roll Call, Inc. | Getty Images
Pfizer said Tuesday it asked the Food and Drug Administration to authorize booster shots of its Covid-19 vaccine for all Americans age 18 and older, a move that would significantly increase the number of people eligible to get the extra shots.
A third dose of the vaccine, which was developed with German partner BioNTech, has already been authorized by the FDA for elderly people and at-risk adults six months after they complete their primary series of shots.
The company said it is asking the agency to amend its authorization after data shows an additional dose is safe and effective in all adults.
This is a developing story. Please check back for updates.
This article was originally published by Cnbc.com. Read the original article here.